New classification PBT and vPvB
There are new classifications in the environmental field that provide a statement about particularly persistent substances since 20.04.2023 in the European Union: PBT and vPvB. The hazard class is called "persistent, bioaccumulative and toxic
New classification endocrine disruption (environment)
Endocrine disruption, i.e. the new classification of hormone-like substances, distinguishes between the disruption for the environment and for human health. In this article, I describe the classification for the effect on the environment. Where
New classification endocrine disruption (human health)
Just after the specification of endocrine disruptors in safety data sheets (article currently only available in German) under REACH, new classifications were presented in the CLP Regulation. In this article, I summarise the classification
CLP Regulation: New classifications are here!
Preliminary versions of the CLP Regulation have been available for some time, and since 31 March 2023 it is official: new classifications are coming in Europe [1]. We will go through the most important key
The tasks of a hazardous substances advisor
In most companies, there are one or more persons who are appointed as hazardous substance advisors. The tasks of a hazardous substance advisor are varied and extensive. In this blog post, I would like to
Acceptance and tolerance concentrations of carcinogenic substances
After more than 10 years since the last revision, an amendment to the Ordinance on Hazardous Substances (GefStoffV) [1] is due this year. I have already dealt with this topic in another blog post. Among
Are your hazardous substances up to date?
In my last blog post, I dealt with the amendment of the Gefahrstoffverordnung (Ordinance on Hazardous Substances) [1]. However, this is not the only legal text that will change in the near future and is
New SDS in 2023
Some people working with SDSs may not have noticed it yet, but we are currently in the transition period to a new SDS format. For the second time, Annex II of the REACH Regulation
The amendment of the Hazardous Substances Ordinance
An amendment to the Hazardous Substances Ordinance (GefStoffV) [1] is due before the end of the year. The Hazardous Substances Ordinance itself has already been amended more frequently, for example in 2010 by adapting it
Effort for the maintenance of a hazardous substances register
If a company uses hazardous substances, these must be documented and managed in a hazardous substances register. The Hazardous Substances Ordinance [1] states that the employer must keep this register, must note certain information and
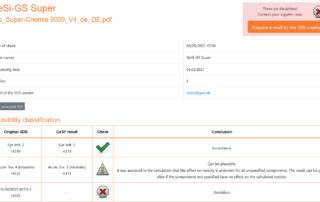
Deviations that aren’t any – the solution lies in the detail
You already know our online tool SDBcheck® (Currently only available in German) for checking the plausibility of your safety data sheets from my last blog posts. Today I would like to discuss the situations in
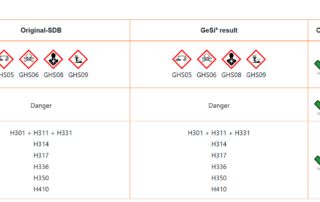
Deviations in the safety data sheet
In my last blog post I dealt with why the plausibility check of safety data sheets is so important (and also obligatory in Germany according to TRGS 400 [1]). With our free online tool SDBcheck®
Plausibility check of safety data sheets
According to the German TRGS 400 [1], you as the recipient of safety data sheets are obliged to check it for "obviously incomplete, contradictory or incorrect information". If you find a discrepancy during the plausibility
What ist the meaning behind product identifiers?
Product identifiers can be found in every safety data sheet - you can' t do without them. But do you know what is actually hidden behind this term? Quite a few legal cross-references in
Help for substitution – the TRGS 600
I have already presented the column model of TRGS 600 [1] to you in recent weeks. However, the TRGS 600 naturally also offers further information and assistance on the subject of substitution. I would
The Column Model – Procedure (Part 7)
In the blog posts of the last few weeks, I have presented the column model of TRGS 600 [1] to you in detail. Now only one more column remains, which I would like to
Software support for SDS and PCN for distributors of hazardous substances
The challenges for distributors of hazardous substances under REACH in the European Union are mostly not apparent through direct obligations. For example, in contrast to the manufacturer/importer, a distributor does not have to meet
The Column Model – Release behaviour (Part 6)
In today's blog post, I would like to introduce you to another column of the column model of TRGS 600 [1]. After the last blog in the series, in which I dealt with column
The Column Model – Physico-Chemical Effects (Part 5)
The column model of TRGS 600 [1] as an assistance for the substitution check is already explained in several blog posts. I already introduced the columns dealing with health and environmental hazards. We now
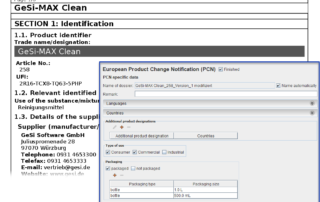
PCN and SDS creation go hand in hand
Both the European Product Notification (PCN) and the Safety Data Sheet (SDS) provide information on hazards of dangerous substances. But they do this with different objectives. Nevertheless, there are many similarities, which is why